#Nonwovens / Technical Textiles
Portable medicines on the rise at Trelleborg
Wearable medical devices are ushering in a future where patient’s diagnosis is more accurate, they can receive tailored treatment when it matters most, and have continuous monitoring of their condition; all with fewer visits to a doctor.
That is why the global wearable medical device market size is projected to grow to 56.6 billion USD by 2030, from 17.4 billion USD in 2020, at a compound annual growth rate of 19.5 percent, according to Market Watch.
Technically, wearable medical devices are nothing new — consumers have been wearing nicotine patches since the 1980s — but a new generation of smart wearables is emerging, including implantable devices, which not only treat a condition but gather huge amounts of health data as well. These devices provide a reliable means of drug therapy without requiring administration by trained personnel and at the same time use sensors to allow doctors to monitor vital signs and parameters such as glucose levels and physical activities in real time.
Wearable devices “enable a more patient-centric approach to diagnostics and therapies versus the way it was done before, where you’d go to the doctor’s office, get a diagnosis, then a prescription,” says Nollenberger. Often these devices “are collecting data in real time, not once a month or however often you go to the doctor. That means earlier diagnoses and more preventative measures that lower overall healthcare costs.”
The technology for wearables is developing rapidly, resulting in designs that are increasingly complex. Manufacturers are challenged to produce devices and components that are implantable and can fit into a 5 mm³ space with a weight of less than 0.02 grams. Drug delivery is also moving through a period of rapid development. Many drugs are being incorporated into small, silicone-based delivery devices, which can release a drug directly with fewer side-effects.
These devices must tick a lot of boxes. Demand is soaring for biocompatible, implantable and bioresorbable material solutions that have functionalities such as conductivity, tracing, adhesion, active pharmaceutical ingredient (API) absorption or lubricity. Wearables that meet the required thresholds must reflect patients’ needs, maximizing comfort and ease of mobility in daily life, all while being easy to use.
“The key to success is design,” says Nollenberger. “The best devices connect a brilliant idea with what’s possible. However, questions require answering; like how can the idea adapt to serial production, rather than be just a one off? Is it scalable to high volume manufacturing? Is it affordable? Is it robust? And ultimately, does it best serve the purpose needed?”
Advances are not governed solely by what is possible. Regulatory hurdles are significant. Cyber security, for example, has become a point of contention. The relatively novel nature of the sector means the regulatory environment is shifting continually, with new demands governing everything from the biological risk of devices to control of standards for chemical substances. The stakes are high, says Nollenberger.
“If you’re wearing something that monitors your blood glucose levels, it has to function,” she adds. “People’s lives are at stake.”
Many obstacles can be overcome when device designers partner with experienced healthcare and medical component manufacturers early in the process. That way, early-stage designs can incorporate expert input on all aspects of the engineering and production process, including prototyping, product and process development, material selection, processing, toolmaking, quality, regulatory and validation planning, scale-up, and automation.
With the right partnerships in place, there is little to stop the proliferation of devices that will further improve patient diagnostics and therapeutics. The pace of change relies on the speed at which inventors can turn academic ideas into functional, scalable devices, according to Nollenberger.
“The research at an academic level is already there in many instances. It’s now a question of people taking that and applying it,” she adds. “Turning that research into usable devices is really about connecting the dots.”
How it works – automated dosing of insulin
Modern diabetes care encompasses fully automated, real-time continuous glucose monitoring (CGM) via wearable or implantable devices combined with a smart insulin pump system.
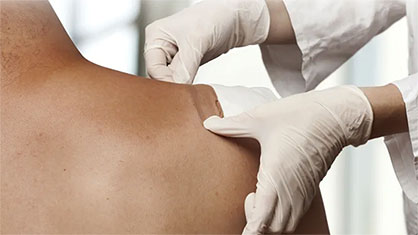
Palm-sized devices attach directly to a patient’s skin with an adhesive patch. Connected to the pump of the device is an extremely fine needle port that extends automatically to inject a pre-programmed quantity of medicine with no input from the user.
Precise control regulates delivery of as little as a few microliters of an active ingredient over a period of time tailored to the particular patient need. This improves the quality of life of diabetics, making delivery of medication flexible and worry-free.
To find out please visit:
https://www.trelleborg.com/en/healthcare














